The Newly Discovered Lactobacillus Plantarum JX183220 uses Cassava (Manihot Esculenta Krantz) Flour
DOI:
https://doi.org/10.31033/abjar.2.1.4Keywords:
lactobacillus plantarum, cassava, flour, box-behnken, fermentation, lactic acidAbstract
The purpose of the current study was to use Response Surface Methodology to optimize the generation of lactic acid using a new isolate of Lactobacillus plantarum JX183220 and cassava flour (Manihot esculenta Crantz) in semi-solid fermentation.
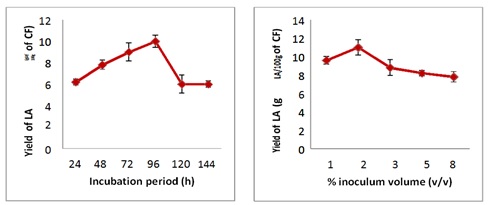
Methodology's Box-Behnken study design was employed. Cassava flour (CF) was employed in a semi-solid fermentation with Lactobacillus plantarum JX183220, a bacteria isolated from goat milk. Initially, preliminary research adjusted a variety of fermentation parameters, including incubation time, inoculum volume, pH, temperature, substrate concentration (cassava flour), and calcium carbonate concentration. The response surface methodology's Box-Behnken design was used to further optimize the substrate concentration, temperature, and pH as potential factors. The fitting of a second-order polynomial regression model, which had a high coefficient of determination, R2, was successful (0.9913). The validation experiment was conducted under the parameters' ideal conditions as determined by the model. According to the preliminary research, Lactobacillus plantarum JX183220 produced the most lactic acid on the fourth day of incubation with 2% inoculum and 0.3% calcium carbonate. Maximum lactic acid production of 18.3679 g/100 g of cassava was achieved by optimization using the Box-Behnken design of RSM at the ideal substrate concentration, temperature, and pH conditions of 36.39°C and 6.43. A validation experiment supported these findings. The best conditions for a newly isolated strain of Lactobacillus plantarum JX183220 to directly convert cassava flour starch to lactic acid were identified. With 15 runs, the RSM's Box Behnken design was determined to be an efficient instrument for maximizing the production of lactic acid. In the future, scientists may use fermentation and sugar-making to make even more lactic acid.
Downloads
References
Adnan AFM, & Tan IKP. (2007). Isolation of lactic acid bacteria from Malaysian foods and assessment of the isolates for industrial potential. Bioresour Technol., 98. 1380-1385.
Sreenath HK, Moldes AB, Koegel RG, & Straub RJ. (2001). Lactic acid production by Simultaneoussaccharificationand fermentation of alfalfa fiber. J Biosci Bioeng., 92(6), 518-523.
Shen XL, & Xia LM. (2006). Lactic acid production from cellulosic material by synergetic hydrolysis and fermentation. Appl Biochem Biotechnol., 133, 251-262.
John RP, Nampoothiri KM, & Pandey A. (2006). Saccharification and fermentation of cassava bagasse for L- (+)-lactic acid production using Lactobacilli. Appl Biochem Biotechnol. 34(3), 263-272.
Naveena BJ, Altaf Md, Bhadrayya K, & Gopal Reddy. (2004). Production of L (+) lactic acid by lactobacillus amylophilusGV6 in semi-solid state fermentation using wheat bran. Food Technol Biotechnol., 42(3), 147–152.
Marques S, Santvos JAL, Gario FM, & Roseiro JC. (2008). Lactic acid production from recycled paper sludge by simultaneous saccharification and fermentation. Biochem Eng J., 41, 210-216.
Giraud E, Gosselin B, Harin B, Parida L, & Raimbault M. (1993). Purification and characterization of an extracellular amylase activity from Lactobacillus plantarum strain A6. J Appl Bacteriol., 75, 276-282.
Naveena BJ, AltafMd, Bhadrayya K, & Gopal Reddy. (2005). Screening and interaction effects of physical parameters, total N content and buffer on l (+) Lactic Acid production in SSF by lactobacillus amylophillus GV6 using Taguchi Designs. Indian J Biotechnol., 4, 342-346.
Hofvendahl K, & Hahn-Hagerdal B. (2000). Factors affecting the fermentative lactic acid production from renewable resources. Enzyme Microb Technol., 26, 87-107.
Anuradha R, Suresh AK, & Venkatesh KV. (1999). Simultaneous saccharification and fermentation of starch to lactic acid. Proc Biochem., 35, 367-375.
Altaf Md, Venkateshwar M, Srijana M, & Reddy G. (206). An economic approach for L (+) lactic acid fermentation by Lactobacillus amylophilus GV6 using inexpensive carbon and nitrogen sources. J Appl Microb., 372-380.
Ramesh RC, Sabita, M, Panda S, & Kar S. (2008). Solid substrate fermentation of cassava fibrous residue for production of α-amylase, lactic acid and ethanol. J Environ Biol., 29(1), 111-115.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2023 Dr. Rajeev Kumar

This work is licensed under a Creative Commons Attribution 4.0 International License.
Research Articles in 'Applied Science and Biotechnology Journal for Advanced Research' are Open Access articles published under the Creative Commons CC BY License Creative Commons Attribution 4.0 International License http://creativecommons.org/licenses/by/4.0/. This license allows you to share – copy and redistribute the material in any medium or format. Adapt – remix, transform, and build upon the material for any purpose, even commercially.


